통합 검색
통합 검색
Device System
To convert carbon dioxide into high-value compounds using electrical energy, device-ization technology is needed to apply the catalyst to actual devices.
Our lab has applied the electrode materials we developed to large-area stack reactors, expanding research beyond laboratory scale to pilot-level systems.
Electrochemical Devices
An electrochemical device is a cell capable of either generating electrical
energy from chemical reactions or using electrical energy to cause
chemical reactions. The electrochemical device which generate an electric
current are called voltaic cells or galvanic cells and those that generate
chemical reactions, via electrolysis for example, are called electrolytic cells.
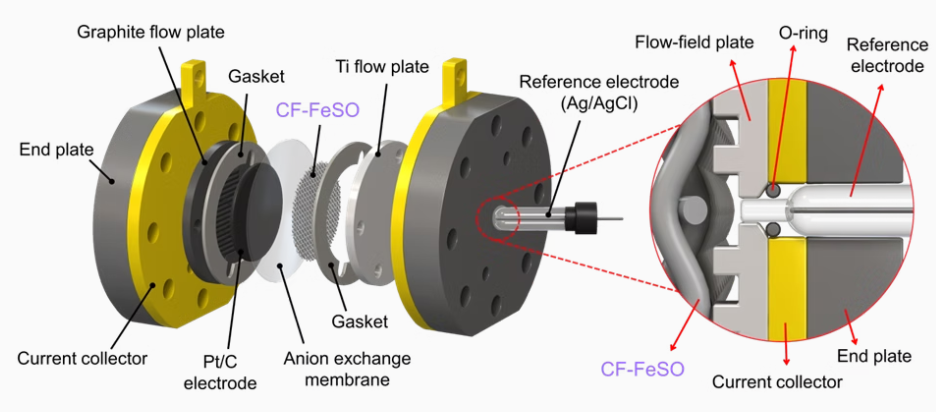
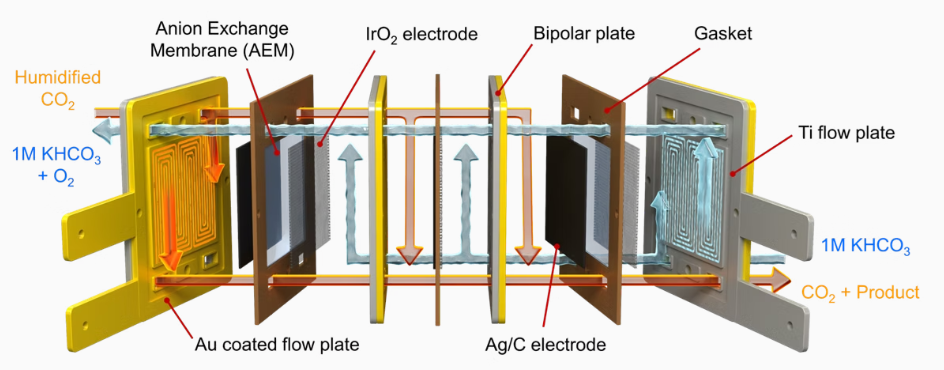
Stack System for CO₂
Reduction Reaction (CO₂RR)
Electrochemical reduction of carbon dioxide represents a possible means
of producing chemicals or fuels, converting carbon dioxide(CO₂) to organic
feedstocks such as formic acid(HCOOH), methanol (CH3OH),
ethylene(C₂H4), methane(CH4), and carbon monoxide(CO). In our lab,
electrode and cell design was researched to produce large amount of
valuable chemicals.
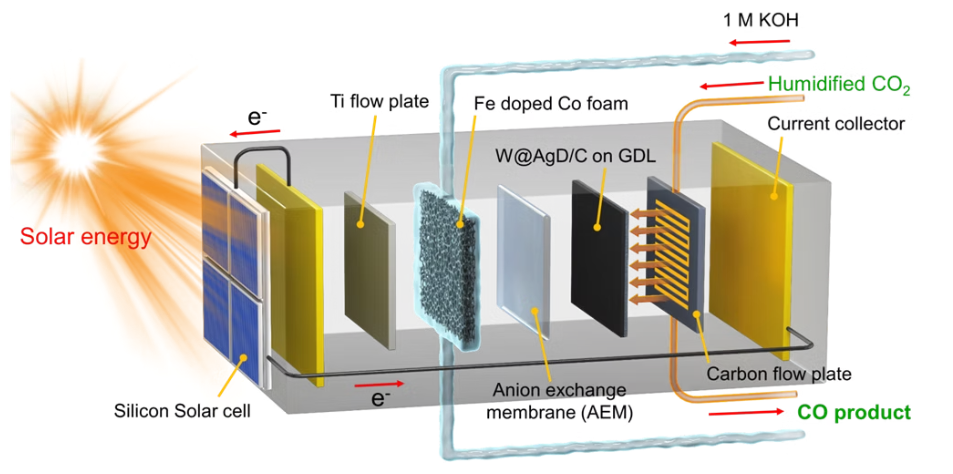
Photo Electrochemical Devices
Solar to fuel is a synthetic chemical fuel produced directly/indirectly from
solar energy sunlight/solar heat through photochemical/photobiological,
thermochemical(i.e., through the use of solar heat supplied by concentrated
solar thermal energy to drive a chemical reaction), and electrochemical
reaction. Light is used as an energy source, with solar energy being
transduced to chemical energy, typically by reducing protons to hydrogen, or carbon dioxide to organic compounds. In our lab, electrode and cell
design was investigated for increasing solar to fuel efficiency.
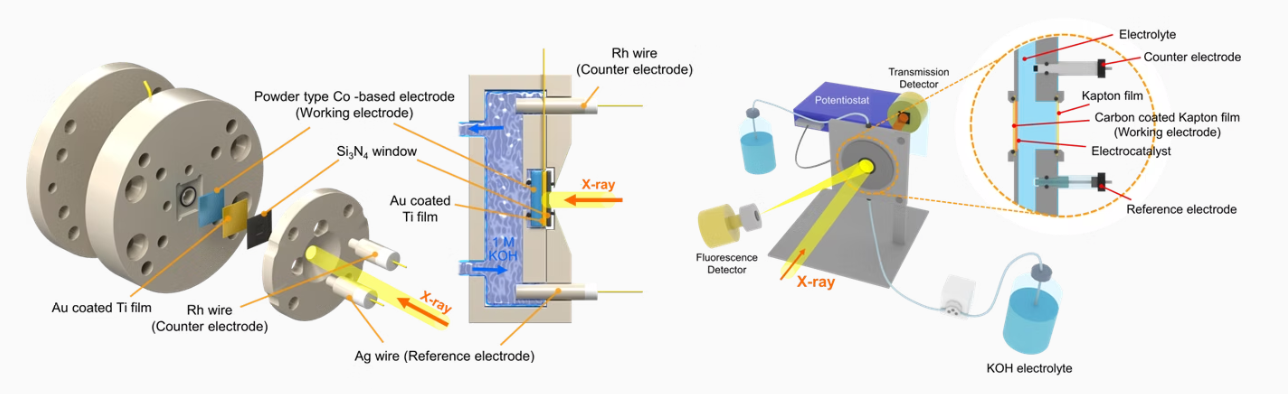
Electrochemical Flow Cell for
in-situ/Operando Analysis
In-situ/Operando analysis is an analytical methodology wherein the
spectroscopic characterization of materials undergoing reaction is coupled simultaneously with measurement of catalytic activity and selectivity.
The primary concern of this methodology is to establish
structure-reactivity/selectivity relationships of catalysts and thereby yield
information about mechanisms. Other uses include those in engineering
improvements to existing catalytic materials and processes and in
developing new ones. In situ/operando analysis requires measurement of
the catalyst under (ideally) real working conditions, involving comparable
temperature and pressure environments to those of industrially catalyzed
reactions. In our lab, various In situ/operando analysis was prepared for
mechanism and characterize study using techniques such as XRD, ICP,
XAFS, and Raman.
.png)
.png)


