통합 검색
통합 검색
About Laboratory
------- Welcome to EEML -------
The lab originated from Yonsei University's Department of Energy and Environmental Engineering.
We are working on various electrochemical reaction systems and materials along with renewable energy
linked sustainable carbon utilization technologies.
We are periodically recruiting graduate students who are interested in these studies and will study together.
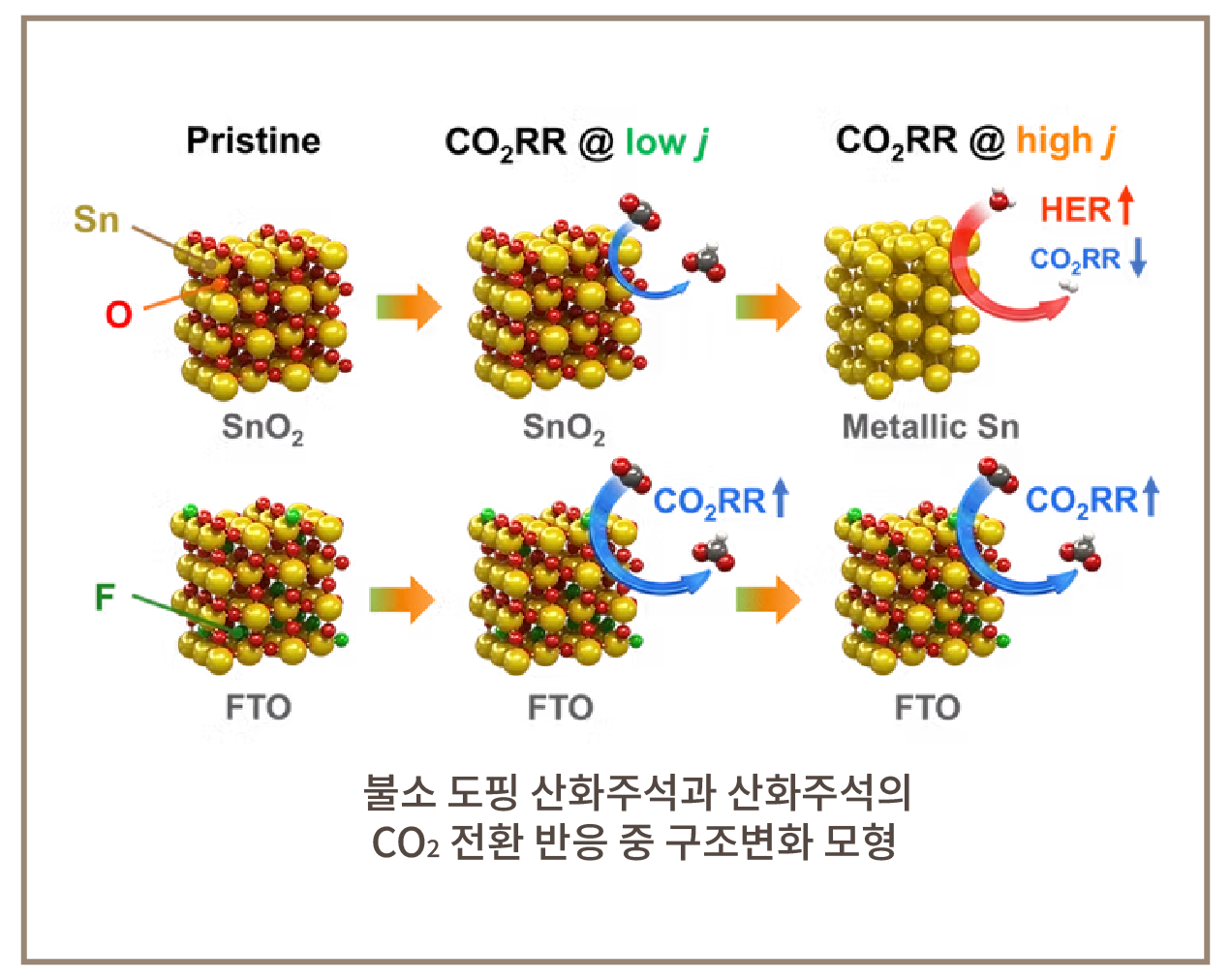
Catalyst Synthesis
Development of various
electrochemical electrode materials
① Nanomaterials: atomic-level, porous,
two-dimensional materials
Nanomaterials can be used safely in
most environments, including
electronic devices that generate
significant heat.
② Dry process electrode material: film
electrode
Research on solid-state batteries and
emerging chemical compositions such
as aluminum and sodium ions.
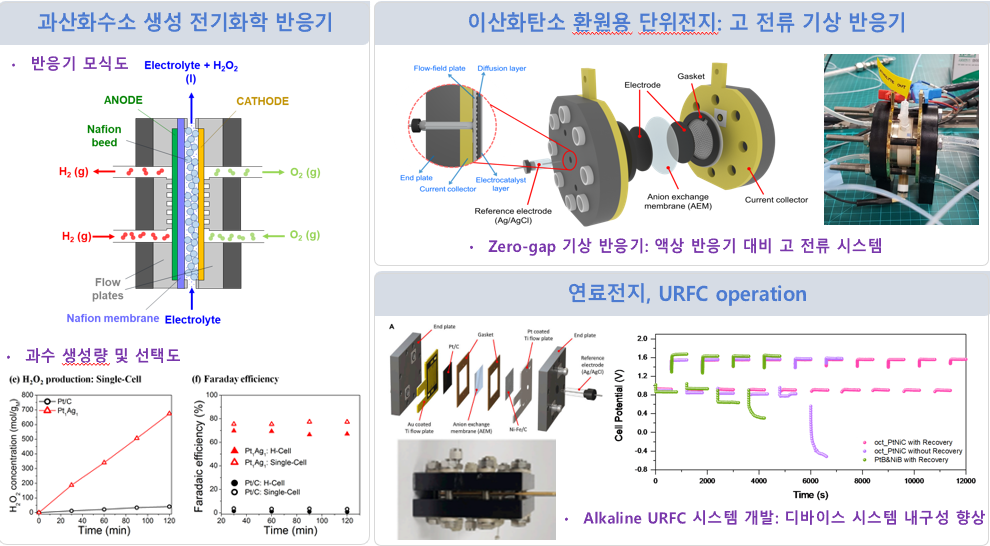
Device System
Electrochemical Flow Cell
for in-situ/Operando Analysis
① Electrochemical reactor
for hydrogen peroxide generation
② Unit cell for carbon dioxide reduction
③ Fuel cell, URFC operation
The flow cell increases the reaction rate
through continuous electrolyte supply
and prevents accumulation of products
or reactants, enabling stable
measurements.
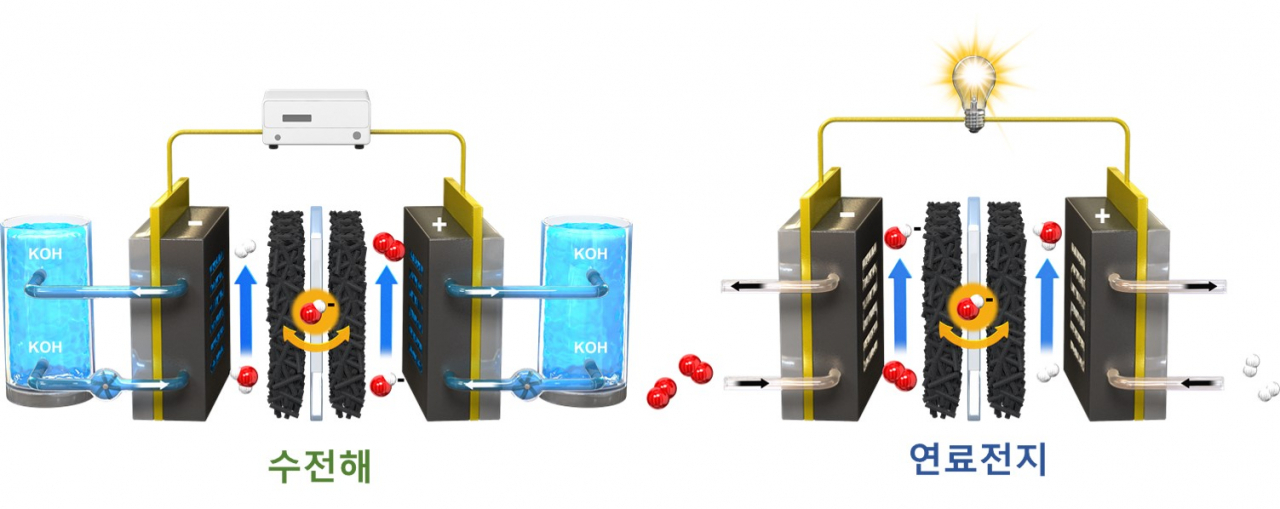
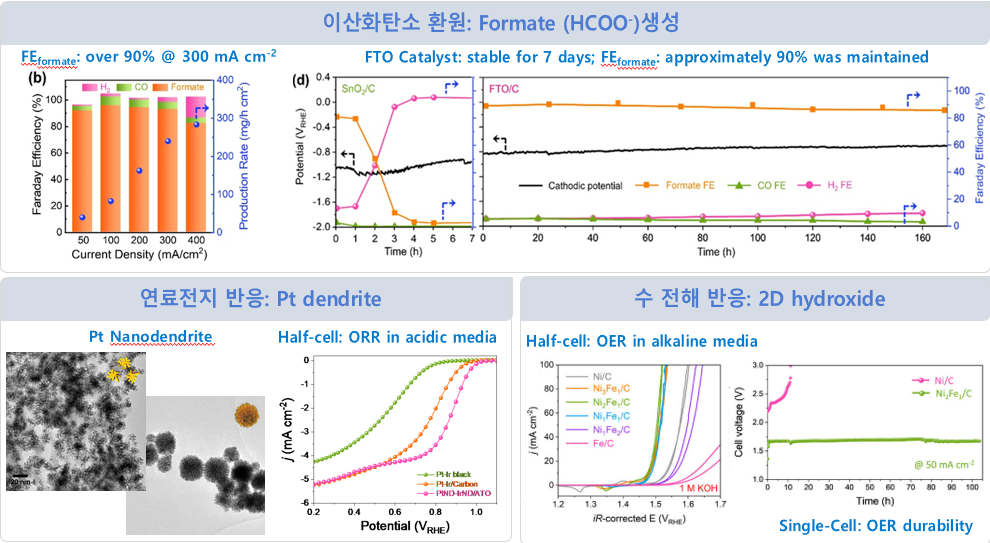
CCUS & Hydrogen Energy
Conducting research
on various electrochemical reactions
① Carbon dioxide reduction
② Fuel cell reaction
③ Water electrolysis reaction
The research is based on the principles
of electrochemistry, which induces
chemical changes by controlling the
oxidation-reduction reaction
of a substance through the transfer
of electrons between an electrode and
an electrolyte.
.png)
.png)



.png)


